What is R4-RA?
A randomised controlled clinical trial in order to determine the response, relapse and resistance to Rituximab therapy (R4) in patients with Rheumatoid arthritis (RA)
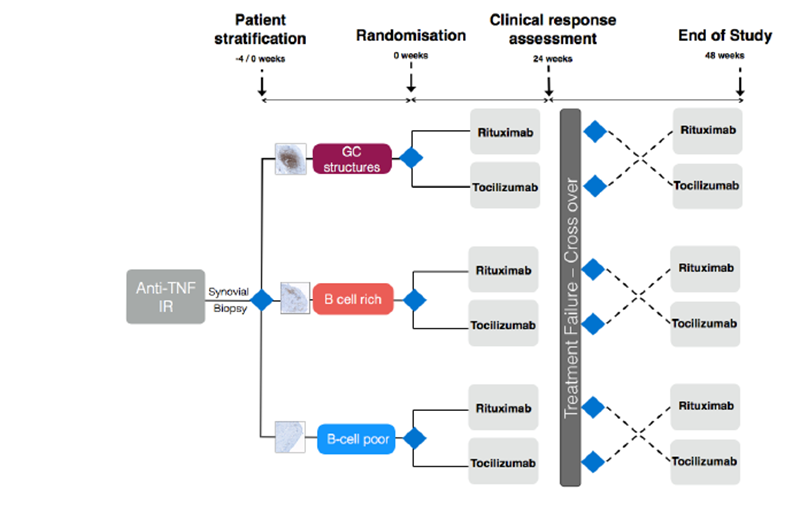
This is a open labelled, randomised clinical trial in anti-TNF alpha inadequate responders to investigate the mechanisms of response, resistance and relapse to Rituximab (R4) versus Tocilizumab in Rheumatoid arthritis (RA): developing a novel biopsy-based diagnostic tool for patient stratification. Patients who fail to respond to anti-TNF alpha drugs will be recruited to this study. They will undergo a synovial biopsy at baseline, prior to randomisation. Patients will subsequently be stratified in to 3 groups (B Cell Poor, B Cell Rich, Germinal Centres (GC) Rich) according to the following B-cell score prior to therapeutic intervention. They will then be randomized to receive Rituximab or Tocilizumab
How many patients will be needed?
We aim to recruit 180 patients over a period of 33 month period.
What are the outcome measures of this study?
The primary outcome measure is the mean change in the clinical disease activity index (CDAI) score at 4 months compared to baseline.
Secondary outcome measures include:
- Clinical Outcomes: the change in the disease activity score (DAS-28), percentage of patients with low disease activity or in remission at 3,6,9 and 12 months
- Radiographic Outcomes: changes in erosive scores using the van der Heijde / Sharp scoring system, changes in US 2d greyscale and power Doppler signal.
- Quality of life outcomes: changes in HAQ / SF-36 scores at 4 and 12 months
In addition to the clinical outcome measures, this study also has exploratory outcomes which will investigate whether Rituximab therapy modulates synovial ectopic lymphoneogenesis and whether this determines clinical response. We will also investigate cytokine expression, B-cell clonal relationships and associated signatures in order to ascertain markers of response / resistance in the synovium and peripheral blood.